Immunotherapy for Canine Meningioma
By G. Elizabeth Pluhar, D.V.M., Ph.D., Dip ACVS1, John R. Ohlfest, Ph.D.2, and Matthew A. Hunt, M.D.
1Department of Veterinary Clinical Sciences, College of Veterinary Medicine; 2Department of Pediatrics, School of Medicine; 3Department of Neurosurgery, School of Medicine
Meningiomas are the most common brain tumor in dogs. Surgical removal is the mainstay of treatment, and most of these tumors are managed with local therapy. Historically, the survival time of dogs with meningiomas treated with surgery alone has been four and one-half to seven months. Systemic therapies so far have had limited effectiveness. However, based on preclinical data from our research program, we embarked on the development of anti-tumor vaccinations to treat dogs with spontaneous meningiomas after surgical removal.
Thirteen dogs have been treated with surgical removal of the tumor followed by a series of vaccinations made from the dog’s own tumor cells (called an “autologous tumor lysate” vaccine), along with one of two immune stimulators. The vaccines were injected into the skin on the back of the dog’s neck two weeks after surgery at suture removal. A total of six vaccinations were given at two-week intervals. All 13 dogs have completed the vaccination series.
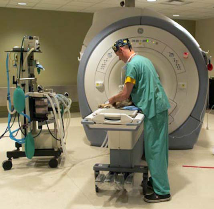
Magnetic resonance imaging (MRI) of the brain was performed immediately after surgery to assess whether the tumor mass was completely removed, and 3, 6, and 12 months after surgery to check for tumor recurrence. Blood collected from each dog during the visits for the MRIs was analyzed for an immune response against the tumor. High-grade malignant tumors were found in two dogs, and low-grade tumors were found in the remaining 11 dogs.
A survival analysis comparing dogs treated with surgery and vaccines to dogs that had surgical removal alone demonstrated a significant survival improvement in the dogs receiving the vaccine therapy. To date, the mean survival time in dogs treated with surgery and vaccines is about one year, with many of the treated dogs still alive and tumor-free, compared to a survival time of approximately 200 days in dogs treated with surgery alone. We also have not seen severe toxicities or side effects in the vaccine-treated dogs. Immune testing demonstrated an antibody response as well as enhanced tumor cell killing by the treated dogs’ lymphocytes.
Our results indicate that immunotherapy using autologous tumor lysate and immune adjuvant after tumor removal elicits specific immune responses against residual tumor cells and increases survival over historical controls. Since this therapy appears to work so well in dogs with meningiomas, it may also be effective in humans with meningiomas that have failed to respond to the standard therapy or are at high risk of recurrence.
Although the pilot phase for this trial is complete and we are not currently recruiting dogs with meningiomas, we are waiting to hear whether two grant proposals to continue our work on dogs with meningiomas will be funded. Check the CVM Clinical Investigation Center website at www.cvm.umn.edu/cic/current/ braintumortrials for more information, applications to enroll dogs in clinical trials, or to make a contribution to support this research.
Help Future Generations of Dogs
Participate in canine health research by providing samples or by enrolling in a clinical trial. Samples are needed from healthy dogs and dogs affected by specific diseases.



